1.It is clarified that the inclusion of the unique traceability code in the packaging of medicines, biological products, and medical devices does not require a modification to their marketing authorization unless changes must be made to the packaging to include said code.
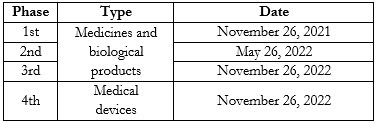
2. The maximum term to implement the traceability of medicines, biological products, and medical devices (hereinafter, the “Products”) is modified, depending on the phase to which they correspond. The national health authority shall determine the Products list for each phase.
3. The members of the National Traceability System (hereinafter, “SNT”) must submit to the National Agency for Sanitary Regulation, Control and Surveillance (hereinafter, “ARCSA”) a gradual plan for the implementation of the traceability of their Products (hereinafter, “Gradual Plan”), according to the regulations issued[1] by ARCSA for this purpose.
4. Until ARCSA has a computer system to control the traceability of the Products, the members of the SNT must record the logistical movements of their Products in the system they have for that purpose.
5. The ARCSA computer system will have a database where all the records generated by the systems of each SNT member will be stored. The characteristics of the ARCSA system shall be indicated in the regulations issued by the said authority for this purpose.
Do you want to receive our newsletters with information like the one you just read?
Click here and subscribe.

